Learn about the advanced form of dry age-related macular degeneration, called geographic atrophy.
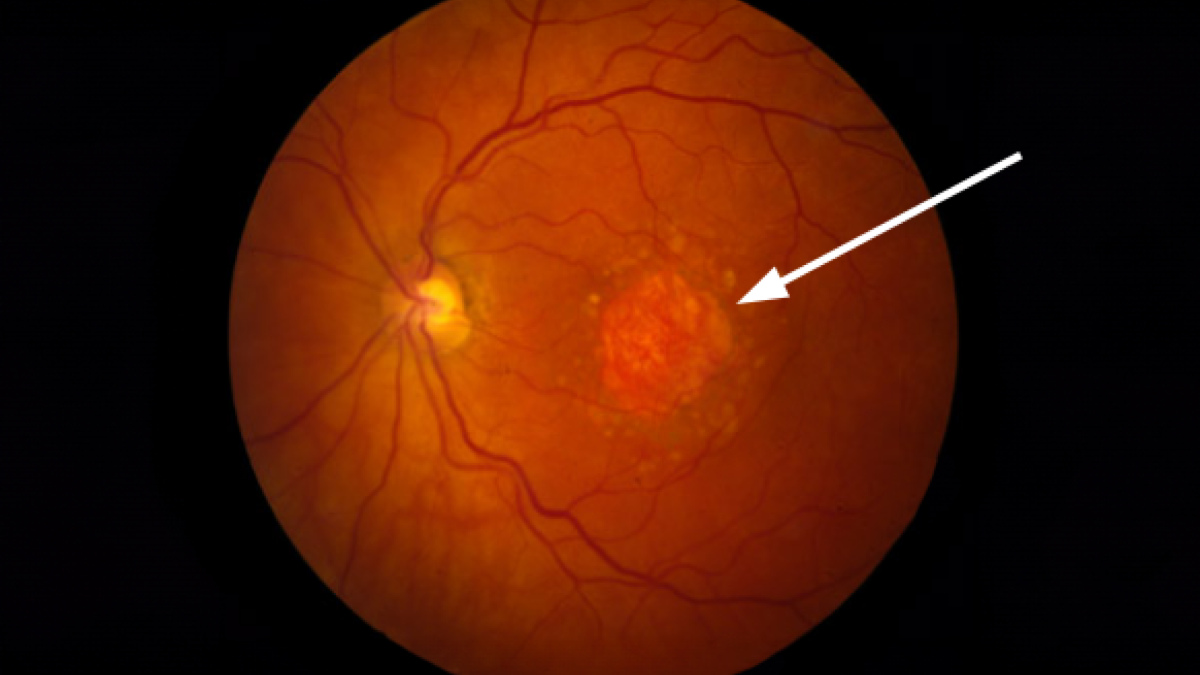
Some patients with age-related macular degeneration (AMD) will develop geographic atrophy, which refers to regions of the retina where cells waste away and die (atrophy). Sometimes these regions of atrophy look like a map to the doctor who is examining the retina, hence the term geographic atrophy. The regions of atrophy result in a blind spot in the visual field. Geographic atrophy can affect one or both eyes, and a patient with geographic atrophy in one eye is more likely to develop it in the other.
Symptoms of Geographic Atrophy
- The initial symptom may be found during reading, when one or several letters in a word are “missing.” Or, when looking at faces, a small part of the face cannot be seen.
- Usually, once geographic atrophy starts, the region of atrophy expands slowly over several years until the central vision is lost and vision is about 20/200. It does not normally affect peripheral vision.
Wet AMD and Geographic Atrophy
Patients with another form of advanced AMD called wet AMD can sometimes also have geographic atrophy before, during, or after they have the wet form of AMD. The two forms of AMD are not mutually exclusive.
Diagnosing Geographic Atrophy
How can you tell if you have geographic atrophy? It can be diagnosed by an ophthalmologist during a dilated exam and/or with retinal imaging. In a dilated exam, geographic atrophy appears as a patch of retina that’s missing its dark melanin pigment. Imaging techniques including retinal color photographs, optical coherence tomography (OCT), or autofluorescence photographs can also be used to detect GA. Read more about the eye exam for macular degeneration.
Treatment
The first treatments for geographic atrophy were approved by the U.S. Food and Drug Administration in 2023. The first, Syfovre, was approved in February 2023, followed by Izervay in September 2023. Both treatments slow the progression of the disease by targeting inflammation in the complement pathway system in the eye. Additional promising treatments are in clinical trials.
Clinical Trials
While a phase III trial by Genentech targeting the complement protein called factor D in geographic atrophy patients was not successful, a phase III trial by Apellis targeting a different complement protein called C3 showed promising results.
The Geographic Atrophy Lipoic Acid (GALA) clinical trial is testing whether a once-per-day antioxidant/iron chelator pill can slow the growth of geographic atrophy.
Additional clinical trials for geographic atrophy can be found at antidote.me or clinicaltrials.gov.
Cell replacement is also being tested for geographic atrophy. A few patients have received stem cell treatments. The most promising of these have the cells grown on plastic sheets, which are surgically placed under the retina and unrolled into position. The cells have been well-tolerated and can survive in the recipient’s eye for at least a year.
Summary
GA robs sight, but advances in low vision therapy (by specially trained optometrists) and implantable miniature telescopes can help patients adapt to their diminished retinal function. New treatments already in clinical trials or being tested in National Institutes of Health and/or foundation-funded labs—like those supported by Macular Degeneration Research, a BrightFocus Foundation program— bring hope for improved vision on the horizon.
About BrightFocus Foundation
BrightFocus Foundation is a premier global nonprofit funder of research to defeat Alzheimer’s, macular degeneration, and glaucoma. Since its inception more than 50 years ago, BrightFocus and its flagship research programs—Alzheimer’s Disease Research, Macular Degeneration Research, and National Glaucoma Research—has awarded more than $330 million in research grants to scientists around the world, catalyzing thousands of scientific breakthroughs, life-enhancing treatments, and diagnostic tools. We also share the latest research findings, expert information, and resources to empower the millions impacted by these devastating diseases. Learn more at brightfocus.org.
Disclaimer: The information provided here is a public service of BrightFocus Foundation and is not intended to constitute medical advice. Please consult your physician for personalized medical, dietary, and/or exercise advice. Any medications or supplements should only be taken under medical supervision. BrightFocus Foundation does not endorse any medical products or therapies.
- Disease Biology
- Dry AMD
- Geographic Atrophy