Key Takeaways
- The use of steroid medications can, in some people, cause elevated pressure within the eye, increasing risk for glaucoma.
- BrightFocus-funded researcher Daniel Stamer, PhD, is using cutting-edge techniques to study the gene Angptl7, which could play a role in that process.
- This work could improve our understanding of how eye pressure is controlled and how it could be targeted to reduce glaucoma risk.
Every day, people take corticosteroids to reduce inflammation, including in their joints, gut, or eyes. They are among the most widely prescribed classes of medications, and their use has become almost an afterthought.
However, in nearly one in three people, corticosteroid use is accompanied by an increase in the eye’s intraocular pressure (IOP)—a primary risk factor for developing glaucoma. As the strength of available corticosteroids continues to increase, so does the potential risk and prevalence of steroid-induced glaucoma.
 Thanks to support from BrightFocus Foundation’s National Glaucoma Research program, Duke University researchers Daniel Stamer, PhD, and Guorong Li, MD, are working to understand the chain of events connecting steroid use and increased IOP.
Thanks to support from BrightFocus Foundation’s National Glaucoma Research program, Duke University researchers Daniel Stamer, PhD, and Guorong Li, MD, are working to understand the chain of events connecting steroid use and increased IOP.
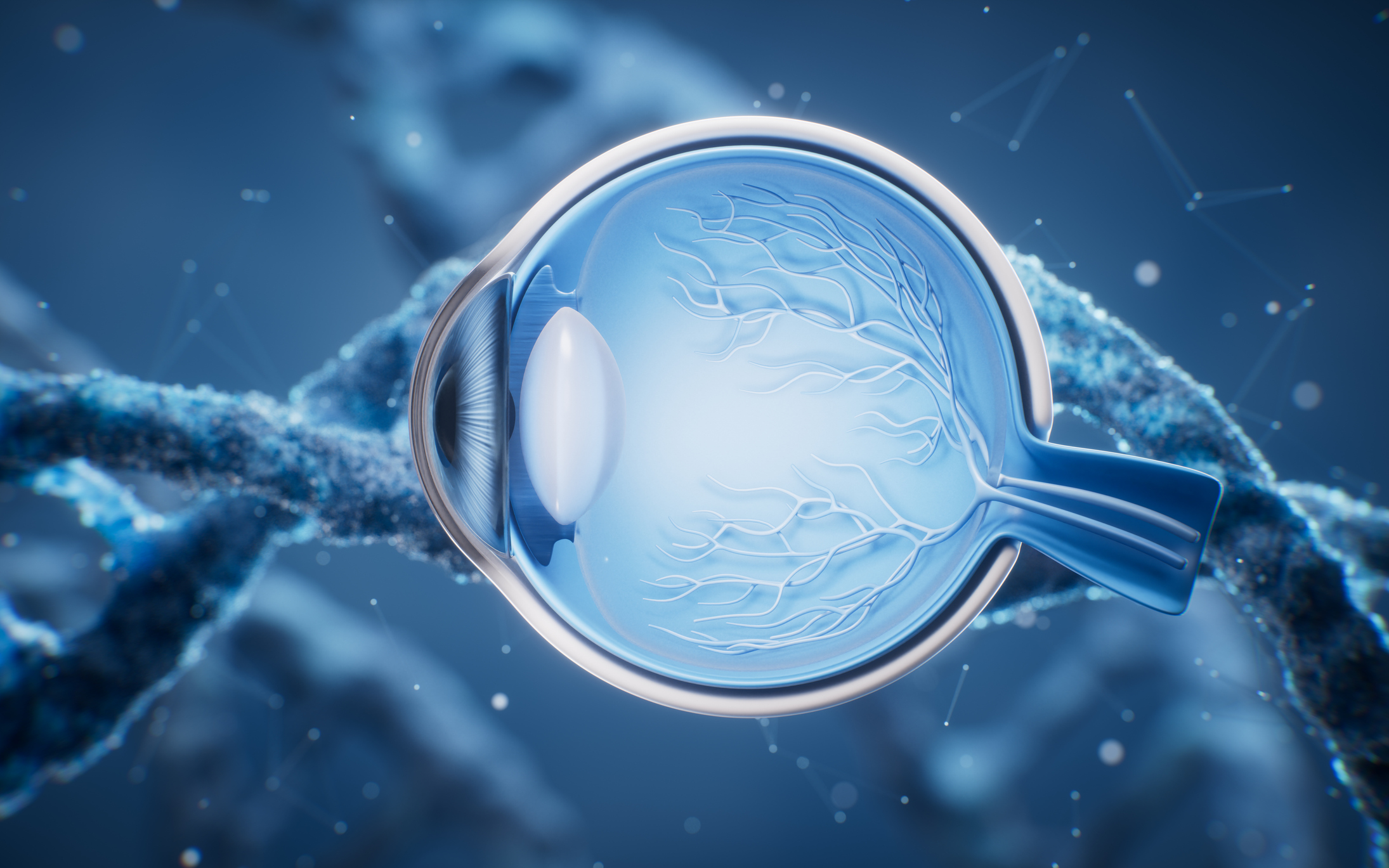
One potential culprit is the gene Angptl7, which can be found within the cells of the trabecular meshwork and Schlemm’s canal, two tissues that make up the eye’s drainage system. Angptl7 has also been linked to glaucoma risk1.
“If you take the gene out of the system, it protects you from steroid-induced glaucoma,” said Dr. Stamer, “so it must have some role, and our project is [trying to] understand what that is.”
Tracking a Silent Culprit
Normally, the aqueous humor found between the cornea and the lens of the eye flows through the trabecular meshworkand Schlemm’s canal into the bloodstream. However, if cells within those tissues malfunction, that fluid flow backs up much like in a clogged sink. With nowhere else to go, the pressure inside the eye increases, which in some cases can lead to glaucoma.
To understand how Angptl7 might be involved in disrupting fluid flow, Drs. Stamer and Li are turning to cutting-edge techniques that let them peer within individual cells inside the eye’s drainage system2. First, they have developed a method for isolating individual, living cells from both the trabecular meshwork and Schlemm’s canal. From there, they apply a technique called singe-cell RNA sequencing, which reveals which genes are actively turned on at any given moment. Together, these methods provide a snapshot of what is happening within cells critical to maintaining IOP.
By combining this with laboratory models that mimic elevated eye pressure, with or without steroid exposure, the researchers can compare what is happening at the cellular level under different conditions. This will allow them to track what changes when IOP increases or decreases and determine what role Angptl7 might be playing in that process.
Previous work has established that, if the function of Angptl7 is suppressed, the eye is protected from developing steroid-induced glaucoma. Teasing out how exactly Angptl7 interacts within and between cells in the eye could lead to potential treatment strategies to prevent an unintended side effect for people who use corticosteroids every day.
Seeding the Science
Dr. Stamer’s research career has spanned 25 years, and his involvement in the glaucoma field was the result of fortuitous happenstance. While working in an eye bank helping obtain tissue for corneal transplant surgeons, he happened to become involved in one of those surgeon’s glaucoma projects.
“At the time I was working at the eye bank, I was introduced to glaucoma because one of the corneal surgeons actually had an idea about how to treat steroid-induced glaucoma,” said Dr. Stamer. “I got very interested in lab work and research in general, and while working on this project, I saw that there was a lot we didn’t know about glaucoma and that it needed someone to spend their career studying how pressure in the eye is controlled.”
He decided to answer that call himself, and National Glaucoma Research was one of the first supporters of his research.
“National Glaucoma Research has been a big part of my career ever since I started my lab 25 years ago,” said Dr. Stamer. “When you are starting out as a researcher and trying to get funding, you have to break the ice somehow, and National Glaucoma Research funding allowed me to generate the preliminary data I needed to apply for bigger grants.”
A quarter century later, that early support is still paying dividends, as Drs. Stamer and Li are at the forefront of research aimed at understanding how IOP is controlled and how it can be treated to reduce the risk of glaucoma. Dr. Stamer emphasized the importance of supporting bold, innovative ideas to both his lab and the field of glaucoma research:
“BrightFocus and its National Glaucoma Research program fill an important niche in the research pipeline that allows scientists to test drive their ideas. This is especially important for people who are still in training, postdoctoral fellows, or people like myself who were just starting their lab.”
From advancing earlier detection to a potential cure, National Glaucoma Research-funded scientists are pushing boundaries thanks to the generous support of people like you. Join the movement to end this sight-stealing disease.




 Thanks to support from BrightFocus Foundation’s National Glaucoma Research program, Duke University researchers Daniel Stamer, PhD, and Guorong Li, MD, are working to understand the chain of events connecting steroid use and increased IOP.
Thanks to support from BrightFocus Foundation’s National Glaucoma Research program, Duke University researchers Daniel Stamer, PhD, and Guorong Li, MD, are working to understand the chain of events connecting steroid use and increased IOP.






