Several hereditary vision diseases affect the macula, the central part of the retina that is responsible for providing sharp, clear, straight-ahead vision. One of them is Best disease, also known as vitelliform macular dystrophy.
Signs & Symptoms
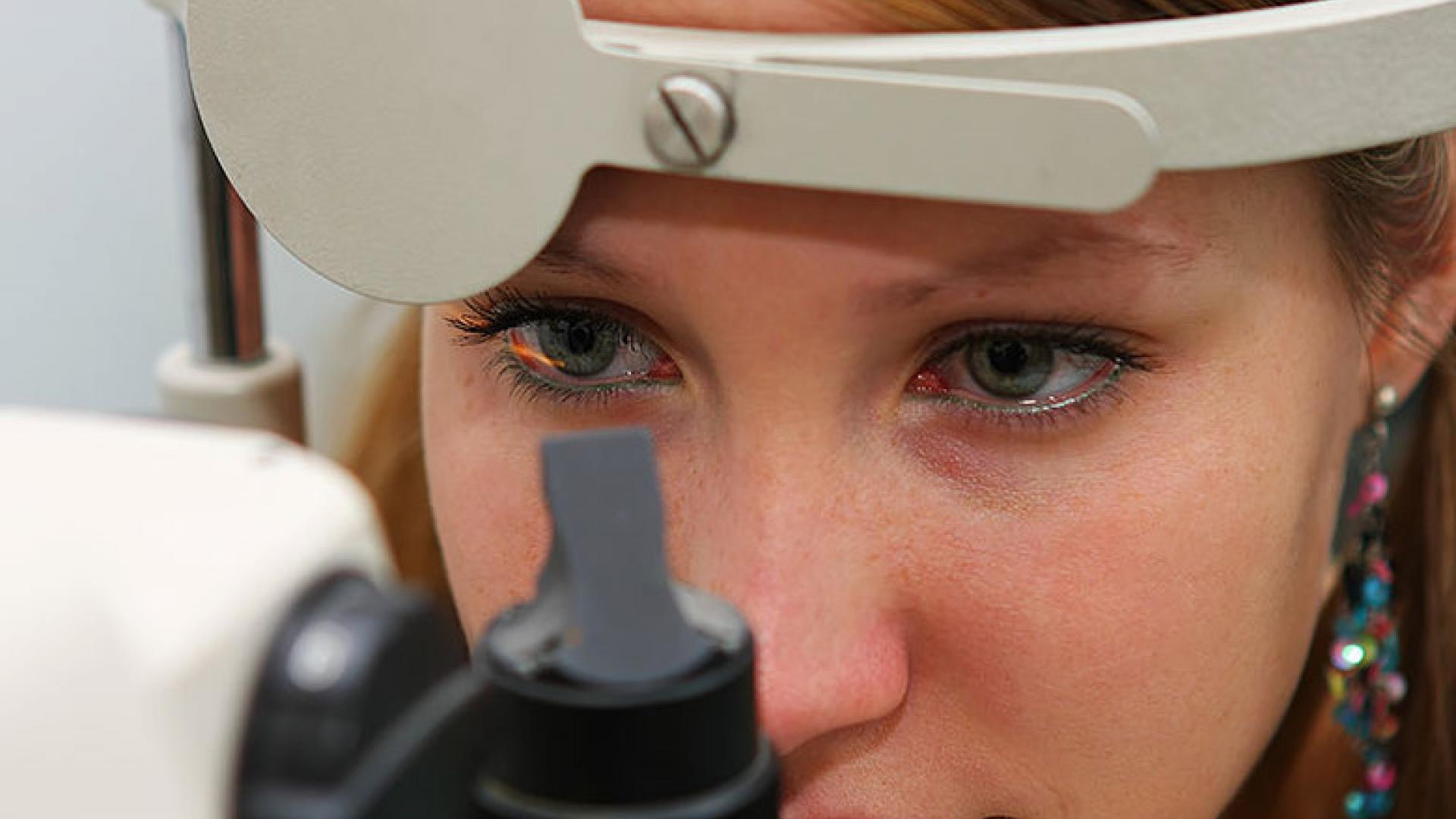
Best disease usually develops in the first decade of life, but may not cause symptoms until later. Symptoms are blurry, distorted, or missing central vision. It can be diagnosed by a dilated eye exam with an ophthalmoscope. In this disease, the macula typically looks like a sunny-side-up egg, which can later burst, releasing fluid into the macula and causing macular degeneration. Additional helpful tests are optical coherence tomography (OCT), a type of noninvasive retinal imaging technique that generates a cross-section picture of the retina in a few seconds, and the electrooculogram (EOG), which measures the electrical potential across the retina.
What Causes Best Disease?
The gene responsible for Best disease is called bestrophin, or vitelliform macular dystrophy 2 (VMD2). Usually, mutations are dominant, so that the children of an affected parent have a 50 percent chance of getting the disease. The normal function of bestrophin is to conduct ions, like calcium and chloride, across the membrane of a cell type in the back of the retina, called the retinal pigment epithelium.
Treatments
Researchers are trying to develop gene and other therapies based on this knowledge, but there are no specific treatments for this disease.
Promising Research
There has been significant progress in research efforts to develop a gene therapy treatment for Best disease. Gene therapy for retinal diseases is now considered a viable approach, as the FDA recently approved retinal gene therapy for an inherited form of blindness called Leber’s congenital amaurosis. The same technical approach, using a non-disease-causing virus called adeno-associated virus (AAV) should now be translatable to additional retinal diseases.
Proof of principle that AAV gene therapy will work for Best disease is provided by the treatment of dogs with Best disease, according to research by investigators at the University of Pennsylvania and the University of Florida.
A company called Ophthotech has a licensing agreement with the University of Pennsylvania to develop these findings into a treatment for patients. Once a clinical trial begins, information on the trial can be found at our Antidote clinical trial search tool or on clinicaltrials.gov.
About BrightFocus Foundation
BrightFocus Foundation is a premier global nonprofit funder of research to defeat Alzheimer’s, macular degeneration, and glaucoma. Since its inception more than 50 years ago, BrightFocus and its flagship research programs—Alzheimer’s Disease Research, Macular Degeneration Research, and National Glaucoma Research—has awarded more than $330 million in research grants to scientists around the world, catalyzing thousands of scientific breakthroughs, life-enhancing treatments, and diagnostic tools. We also share the latest research findings, expert information, and resources to empower the millions impacted by these devastating diseases. Learn more at brightfocus.org.
Disclaimer: The information provided here is a public service of BrightFocus Foundation and is not intended to constitute medical advice. Please consult your physician for personalized medical, dietary, and/or exercise advice. Any medications or supplements should only be taken under medical supervision. BrightFocus Foundation does not endorse any medical products or therapies.
- Treatments