In 2018, the manufacturers of the CyPass micro-stent decided to voluntarily remove it from the market. Learn why they took this step, and what you should do if you have already had the CyPass micro-stent procedure.
What is the CyPass Micro-Stent?
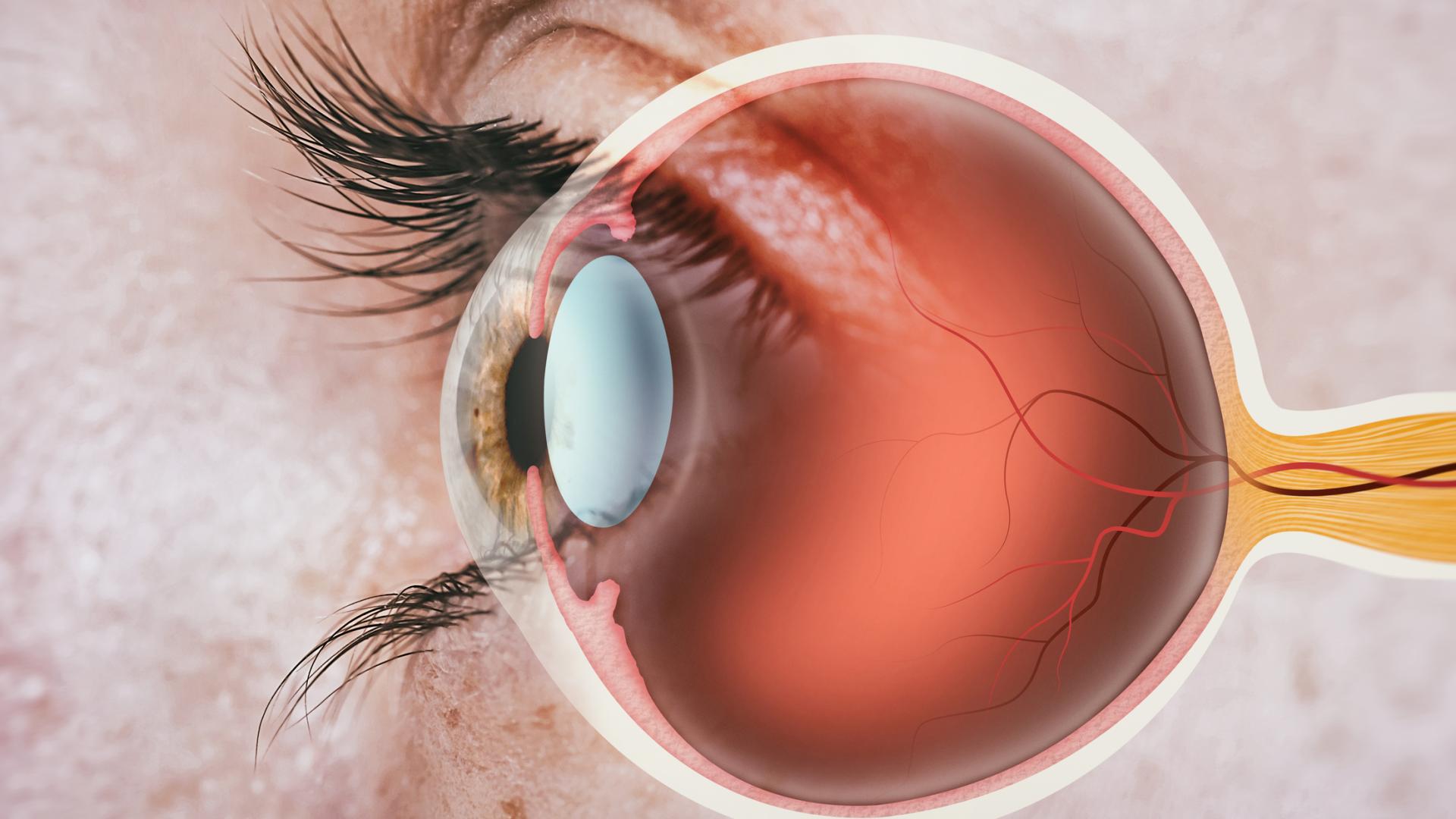
The CyPass micro-stent was FDA approved in 2016 to help lower eye pressure when combined with cataract surgery in patients with mild-to-moderate primary open-angle glaucoma. It is a very small plastic tube implanted in the eye, allowing the fluid to drain and lower the eye pressure. The CyPass is implanted at the time of cataract removal through the same incision your ophthalmologist uses to remove your cataract.
Cataract surgery combined with CyPass lowered eye pressure on average to approximately 16 mmHg (millimeters of mercury) in randomized controlled clinical trials. The FDA approved the device when patients had been followed for two years. At two years, there were no differences in corneal cell loss between patients with cataract surgery combined with CyPass compared to patients who had cataract surgery alone.
Loss of Cornea Cells
When the FDA approved the CyPass in 2016, it also required that the manufacturer follow patients in the clinical trial for an additional three years after surgery (five-year data is now available). It was discovered that at five years, patients with the CyPass device had more loss of special cells, called endothelial cells, in the cornea (the clear window or outer protective layer of the eye) compared to those patients who had cataract surgery alone. It should be noted that cataract surgery alone does lead to a slow rate of endothelial cell loss.
Endothelial cells line the inner surface of the cornea and are critical for pumping fluid out of the cornea and keeping it clear. A clear cornea is important for clear vision. Endothelial cells cannot regenerate, so if they are damaged, they do not regrow. When the cornea loses endothelial cells, it is possible for the cornea to swell and lead to loss of vision. Luckily, unlike glaucoma, which is irreversible, the corneal endothelial cells can be replaced, but it requires a partial corneal transplant surgery.
Voluntary Withdrawal of CyPass
The preliminary data also shows that more endothelial cells were lost when the device was not implanted deep enough and was closer to the cornea. Because of these findings, the manufacturer decided to voluntarily withdraw CyPass from the market. This does not mean that the CyPass should be removed after it has already been implanted, but rather no further CyPass micro-stents will be implanted, and unused devices have been returned to the manufacturer.
Although the five-year data shows that there was greater endothelial cell loss in those patients who had the CyPass, there were no patients who required a corneal transplant due to the CyPass. Of course, we do not know what will happen over the longer term. Thus the most important recommendation to patients with CyPass devices implanted is to continue to follow up with your surgeon. It is not recommended to remove the micro-stent at this time since only a subset of patients demonstrated endothelial cell loss, and at this point, there has not been swelling of the cornea resulting in the need for corneal transplantation among the patients in the clinical trial.
What Should You Do?
If you have had the CyPass implanted, make an appointment with your ophthalmologist, and they will assess your cornea at each visit, in addition to monitoring the position of the CyPass. Some ophthalmologists may also begin monitoring your endothelial cell count and corneal thickness. You and your ophthalmologist should discuss the most appropriate next steps moving forward.
About BrightFocus Foundation
BrightFocus Foundation is a premier global nonprofit funder of research to defeat Alzheimer’s, macular degeneration, and glaucoma. Since its inception more than 50 years ago, BrightFocus and its flagship research programs—Alzheimer’s Disease Research, Macular Degeneration Research, and National Glaucoma Research—has awarded more than $300 million in research grants to scientists around the world, catalyzing thousands of scientific breakthroughs, life-enhancing treatments, and diagnostic tools. We also share the latest research findings, expert information, and resources to empower the millions impacted by these devastating diseases. Learn more at brightfocus.org.
Disclaimer: The information provided here is a public service of BrightFocus Foundation and is not intended to constitute medical advice. Please consult your physician for personalized medical, dietary, and/or exercise advice. Any medications or supplements should only be taken under medical supervision. BrightFocus Foundation does not endorse any medical products or therapies.
- Eye Health
- Treatments