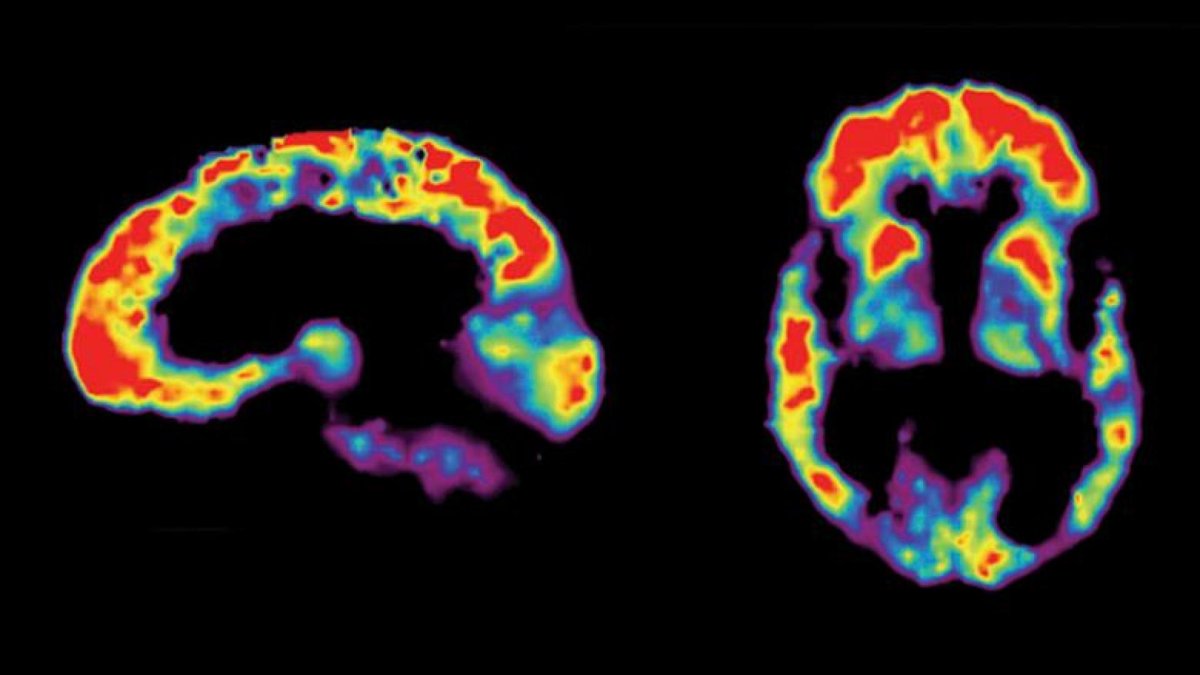
Amyloid PET scans allow, for the first time, accurate detection of amyloid plaques—one of the hallmarks of Alzheimer’s disease—in living people. The scans may help clinicians proceed with greater knowledge and assurance, and patients will be able to have greater confidence in their diagnoses and treatment recommendations. This article explains what PET scans are and how they are currently used, and provides an update on a new clinical trial that will deliver important information on how the scans will be used in the future.
A Crucial Second Opinion
Mr. Swift* arrived for his “second opinion” consultation with the requested folder of medical reports, neuropsychological assessments, lab results, and MRI disks. In his initial meeting with another physician, he had been told that his troubling forgetfulness was due to Alzheimer’s and that this condition would likely progress to disability. He was advised to consider his approaching decline as he and his dear family planned for their future. He began to plan an exit from his thriving business. At age 62, in what he had considered his prime, this was terrifying news.
He wanted a second opinion, and that is how he found me. A careful evaluation of his history, health status, and thorough prior evaluations couldn’t rule out the possibility of Alzheimer’s, although his age of onset was young, and some symptoms were not typical.
After some discussion, we agreed he should have an amyloid PET scan, even though it would have to be purchased without insurance coverage. The reassuring negative scan indicated that he did not have Alzheimer’s or very early Alzheimer’s. The negative scan results indicated that it was important to look for other causes of cognitive difficulty (such as vitamin deficiency or mercury toxicity), which prompted further medical assessment and identified issues that were treated aggressively. Mr. Swift’s cognition improved and stabilized, and several years after these events, he continues to enjoy an independent and active life.
What is PET?
PET, or Positron Imaging Tomography, is a nuclear medicine neuroimaging tool that can be used to make a picture of amyloid plaque accumulation within a person’s brain. Amyloid can accumulate with age in a brain that is functioning normally, and it can be seen in diseases other than Alzheimer’s, but the presence of a positive amyloid scan in a symptomatic individual is a strong indicator of an Alzheimer’s diagnosis. In this type of scan, specially prepared tracer molecules are injected intravenously. They travel through the blood to the brain and attach to amyloid plaques. The radiation they send out can be used to create a picture that tells us the density and location of plaques. Tracers of this type have already been used as research tools, and the FDA has recently approved several for clinical use.
How is PET Used?
Amyloid PET scanning has already revolutionized clinical research. For many clinical drug trials, a positive amyloid scan is now required prior to enrollment. This way, investigators can be more certain they are treating people who truly have Alzheimer’s. When subjects are screened in this way prior to treatment, the effectiveness of a new medication is more likely to be recognized.
The clinical roles for amyloid PET scanning remain much less clear at this time. A group of experts met to discuss this and published their recommendations in 2011. They suggested that these scans should be used clinically when the results would make a difference in treatment. The groups of patients whose treatment might be changed in a helpful way by a PET amyloid scan result include:
- Patients with persistent or progressive and unexplained mild cognitive impairment
- Patients diagnosed with possible AD but with unusual features such as rapid course, unexpected symptoms, or concurrent medical issues
- Patients with unusually early onset of dementia who might benefit from different treatments or from referral to a clinical trial on the basis of a scan result
On the other hand, the consensus group discouraged the use of amyloid imaging for patients whose management would not be likely to benefit, including:
- Patients with typical Alzheimer’s symptoms, age of onset, and course
- Patients without symptoms or whose symptoms are not confirmed by clinical examination
- Patients seeking to know their risk for Alzheimer’s because of various concerns, including a family history of dementia or the presence of a particular version of the ApoE gene, or patients with a strong family history of early-onset Alzheimer’s seeking an alternative to gene testing.
- Individuals being screened for insurance coverage, employment, or other nonmedical purposes
Researchers have already begun to see whether the results of amyloid PET scanning have practical benefits in clinical use. Grundman and colleagues, for example, recently published results from a study that explored how clinicians might change their treatment plans for patients with positive or negative amyloid scan results.
For patients with an Alzheimer’s diagnosis and negative scan, cholinesterase medications (such as Aricept, Exelon, and Razadyne) were typically stopped. These medicines can be expensive and sometimes produce uncomfortable side effects, so it is worthwhile to stop prescribing them to patients who will not benefit. For subjects in this study who had carried a non-Alzheimer’s diagnosis but had a positive scan, the diagnosis was typically changed, and appropriate medications started. Subjects who learned of their scan results were not more likely to become depressed or anxious. In another study that focused on earlier onset subjects, 18% of patients initially thought to have Alzheimer’s turned out to have negative amyloid scans. A negative scan can fuel a more intensive search for treatable causes of cognitive problems, sometimes with results as fortunate as Mr. Swift’s.
A Large Multicenter Trial
The IDEAS study (Imaging Dementia—Evidence from Amyloid Scanning) explored whether results from appropriately ordered amyloid scanning would affect clinical management. Results published in 2019 supported the usefulness of this test. Clinicians changed their management approach often on the basis of scan results, in some cases prescribing Alzheimer’s disease medications earlier and in others abandoning an incorrect initial diagnosis of probable Alzheimer’s disease.
Stay Tuned
For Mr. Swift, amyloid PET scanning was a definite game-changer. So far, it looks like these scans can help others as well. The major drawbacks will be clinically appropriate use, scan availability, and expense, but evidence that scan results affect clinical decision-making seems to be accumulating. Whether positive or negative for amyloid accumulation, these scans may help clinicians proceed with greater knowledge and assurance. Patients will be able to have greater confidence in their diagnoses and treatment recommendations. As the development of disease-modifying treatments becomes ever more commonplace, a tool that increases diagnostic accuracy will be welcome. Stay tuned for new decisions from the Centers for Medicare & Medicaid Services regarding scan coverage by Medicare.
*Mr. Swift’s name and details have been changed to protect confidentiality.
About BrightFocus Foundation
BrightFocus Foundation is a premier global nonprofit funder of research to defeat Alzheimer’s, macular degeneration, and glaucoma. Since its inception more than 50 years ago, BrightFocus and its flagship research programs—Alzheimer’s Disease Research, Macular Degeneration Research, and National Glaucoma Research—has awarded more than $330 million in research grants to scientists around the world, catalyzing thousands of scientific breakthroughs, life-enhancing treatments, and diagnostic tools. We also share the latest research findings, expert information, and resources to empower the millions impacted by these devastating diseases. Learn more at brightfocus.org.
Disclaimer: The information provided here is a public service of BrightFocus Foundation and is not intended to constitute medical advice. Please consult your physician for personalized medical, dietary, and/or exercise advice. Any medications or supplements should only be taken under medical supervision. BrightFocus Foundation does not endorse any medical products or therapies.








